FDA Approves First-Ever Gene Therapy for Genetic Hearing Loss: A Transformational Milestone in Precision Medicine
The recent approval by the U.S. Food and Drug Administration of the first-ever gene therapy designed to treat genetic hearing loss marks a defining moment in the evolution of modern medicine. Granted under the National Priority Voucher Program, this breakthrough is more than a regulatory milestone—it signals the arrival of a new era where previously […]
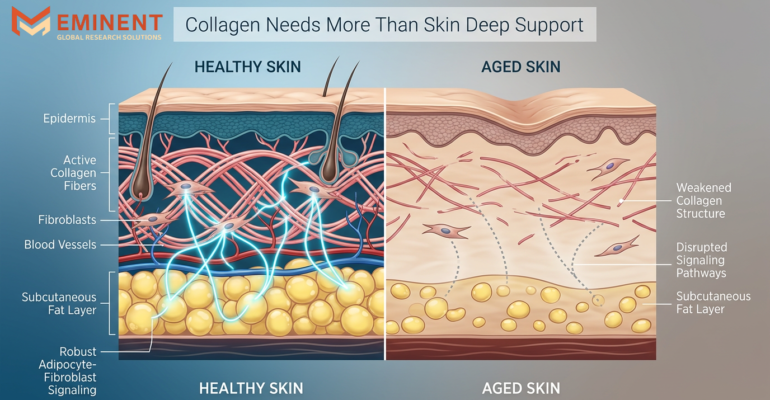
The Silent Collapse: How Loss of Subcutaneous Fat Disrupts Collagen Signaling And Why No Formulation Can Fully Replace It
The Silent Collapse: How Loss of Subcutaneous Fat Disrupts Collagen Signaling And Why No Formulation Can Fully Replace It By Eminent Global Research Solutions The modern skincare and aesthetic industry is built on a powerful promise: that topical formulations, injectables, and advanced treatments can restore youthful skin. While these innovations have delivered impressive results, a […]
The Evolving Landscape of the Cosmetic Industry: Trends, Innovation, and Market Intelligence
The Evolving Landscape of the Cosmetic Industry: Trends, Innovation, and Market Intelligence The global cosmetic industry has transformed dramatically over the past decade, evolving from a product-centric market into a dynamic ecosystem driven by innovation, consumer awareness, and digital influence. Today, cosmetics are no longer limited to beauty enhancement; they represent self-expression, wellness, sustainability, and […]
Big Oil, Smarter Data: How AI Is Redefining the Energy Industry
The global energy sector is undergoing one of the most profound transformations in its history. Once driven primarily by physical assets—rigs, pipelines, refineries—it is now being reshaped by something far less tangible but infinitely powerful: data. Artificial Intelligence (AI) is no longer an experimental tool in oil and gas; it has become a strategic imperative. […]
Digital Infrastructure Funds Are Reshaping Telecom Ownership
Digital Infrastructure Funds Are Reshaping Telecom Ownership The global telecom industry is undergoing a structural transformation, and at the center of this shift is the growing influence of digital infrastructure funds. Traditionally dominated by telecom operators with vertically integrated business models, the sector is now witnessing a steady separation of infrastructure ownership from service delivery. […]
FDA’s Flexible Approach to Cell & Gene Therapy A Strategic Reset for Biopharma
The latest regulatory evolution from the U.S. Food and Drug Administration is not merely about introducing flexibility in cell and gene therapy (CGT) development—it represents a fundamental shift in how innovation will be evaluated, scaled, and commercialized. What appears on the surface as regulatory easing is, in reality, a strategic reset that is redefining competitive […]
Purdue Pharma’s Imminent Sentencing: A Turning Point in the Opioid Crisis and What It Means for Settlement Funds
The long-running legal saga surrounding Purdue Pharma is approaching a critical milestone. With sentencing in the opioid case expected imminently, the path is being cleared for one of the most significant settlement payouts in U.S. legal history to finally begin distribution. If implemented as anticipated, the settlement—approved last year—could take effect as early as May […]
Xenotransplantation Breakthrough: From Scarcity to Scalability in Organ Transplants
The global healthcare ecosystem has long been constrained by one of its most critical bottlenecks: the availability of transplantable human organs. Thousands of patients remain on waiting lists, with many never receiving the life-saving interventions they need. However, a recent breakthrough in xenotransplantation—where a genetically modified pig kidney functioned successfully in a human for 271 […]
FDA’s Regulatory Flexibility in Cell & Gene Therapy A Strategic Inflection Point for Biopharma
The U.S. Food and Drug Administration (FDA) has introduced a more flexible approach to regulating cell and gene therapies (CGTs), particularly in chemistry, manufacturing, and controls (CMC). This shift is not just a regulatory update—it represents a structural change in how innovation is evaluated, accelerated, and commercialized in advanced therapeutics. For companies operating in this […]
Article: Regulatory Easing in Digital Health A Strategic Inflection Point for Innovation and Growth
The evolving regulatory landscape in digital health is entering a pivotal phase. The U.S. Food and Drug Administration (FDA) is signaling a more flexible approach toward low-risk digital health tools, while maintaining rigorous oversight for high-risk, AI-driven clinical systems and software that functions as a medical device. At first glance, this may appear to be […]
Search
Our Projects
Popular Tags
Subscribe Us
Subscribe us & get
latest news & articles to inbox.