Strategic Case Study & Commercial Expansion Framework for Global MedTech Companies
The global MedTech industry is undergoing rapid transformation driven by AI-enabled diagnostics, digital therapeutics, remote monitoring, value-based healthcare, and accelerated regulatory pathways. Companies entering international markets face increasing complexity around reimbursement systems, regulatory compliance, localization, and strategic partnerships.
This case study demonstrates how a MedTech company can successfully prioritize international markets and establish high-value commercialization partnerships using a structured framework focused on:
- Market attractiveness
- Regulatory feasibility
- Partner ecosystem maturity
- Commercial scalability
- Reimbursement readiness
- Competitive positioning
The report also outlines how strategic partner selection can reduce commercialization risk, accelerate market entry timelines, and improve long-term revenue growth.
Industry Background
The global MedTech ecosystem is shifting toward:
- AI-driven healthcare solutions
- Precision diagnostics
- Digital health integration
- Outpatient and ambulatory care expansion
- Regional manufacturing diversification
- Faster commercialization models
According to recent MedTech trend reports:
- Europe represents nearly 27% of global MedTech revenues
- AI-focused MedTech investment continues to grow rapidly
- Regulatory readiness and reimbursement strategy are now major investor evaluation criteria
Business Challenge
A mid-sized MedTech company specializing in digital diagnostics and connected medical devices planned international expansion beyond its domestic market.
Key Challenges
- Selecting the most commercially viable countries
- Managing diverse regulatory requirements
- Identifying reliable distribution and commercialization partners
- Building reimbursement access strategies
- Reducing time-to-market
- Optimizing investment allocation
Strategic Objectives
| Objective | Target Outcome |
|---|---|
| Prioritize high-growth markets | Faster commercialization |
| Build regional partnerships | Reduce operational risk |
| Improve regulatory planning | Accelerate approvals |
| Increase ROI on expansion | Sustainable revenue growth |
| Establish scalable GTM model | Multi-country rollout |
Market Prioritization Framework
The company evaluated international opportunities using a weighted scoring model.
Key Evaluation Parameters
| Criteria | Weight |
|---|---|
| Market Size & Growth | 25% |
| Regulatory Complexity | 20% |
| Reimbursement Potential | 15% |
| Competitive Intensity | 10% |
| Partner Ecosystem | 15% |
| Digital Health Adoption | 15% |
Market Prioritization Results
| Country | Opportunity Score | Commercial Potential |
|---|---|---|
| United States | 92 | Very High |
| Germany | 88 | High |
| UAE | 81 | High |
| Singapore | 79 | Medium-High |
| Saudi Arabia | 77 | Medium-High |
| India | 75 | High Volume |
| Brazil | 69 | Emerging |
Partner Strategy Framework
The company adopted a hybrid commercialization model based on regional market maturity.
Partnership Categories
| Partner Type | Role |
|---|---|
| Regulatory Partners | Approval support |
| Distribution Partners | Market access |
| Clinical Networks | Validation & adoption |
| Reimbursement Advisors | Pricing strategy |
| Local Commercial Teams | Sales enablement |
| Technology Alliances | Product integration |
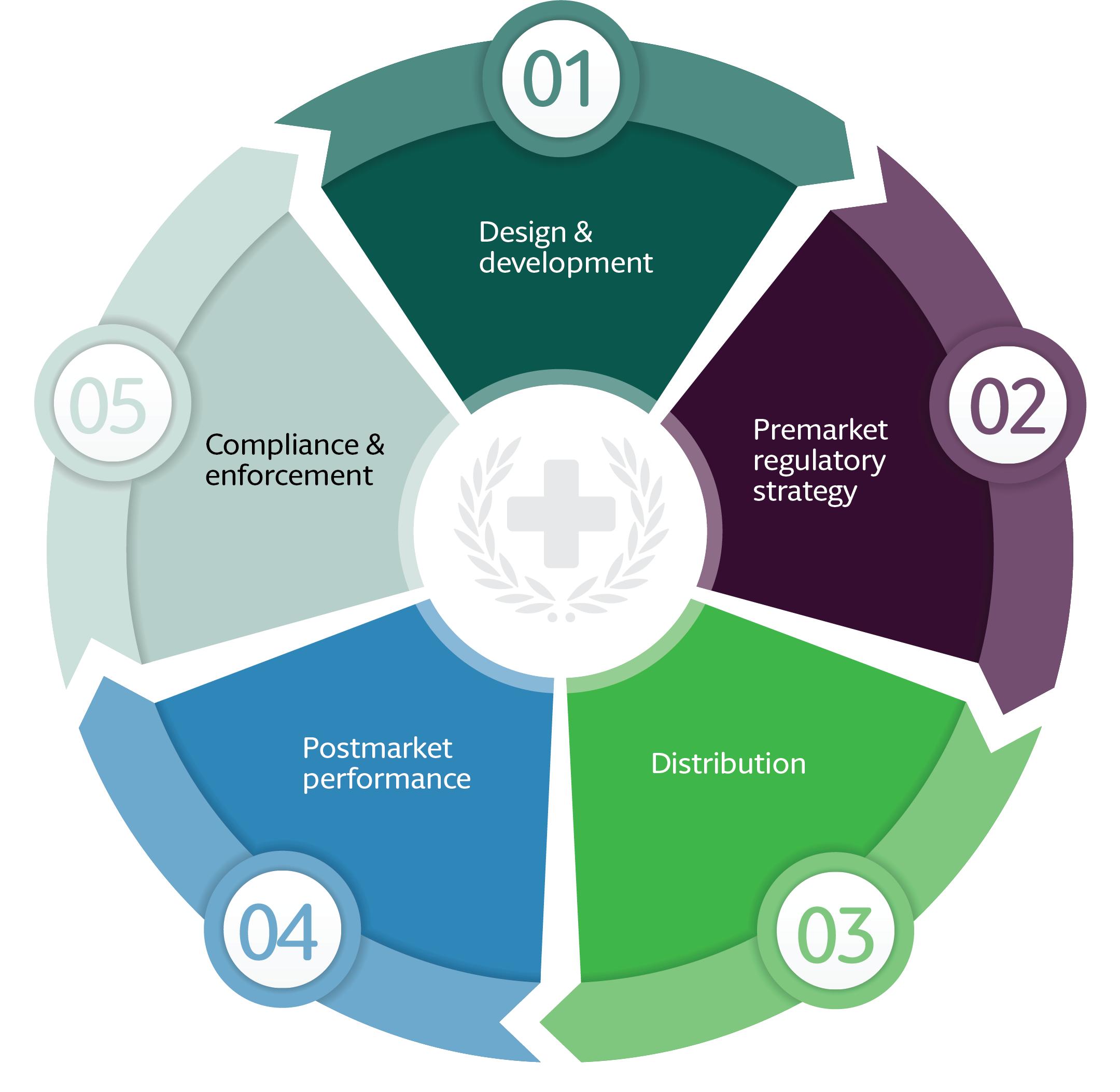
Regulatory Strategy Analysis
Regulatory acceleration became a key commercialization advantage.
The company aligned its regulatory strategy with:
- FDA pathways in the United States
- MDR compliance in Europe
- GCC healthcare standards in the Middle East
- Digital health frameworks in APAC markets
Accelerated approval pathways and early regulatory engagement significantly reduced launch delays.
Financial Impact Assessment
Estimated Outcomes After 36 Months
| KPI | Result |
|---|---|
| Market Entry Time Reduction | 32% |
| Commercialization Cost Reduction | 24% |
| Partner Acquisition Efficiency | 41% |
| International Revenue Growth | 68% |
| Regulatory Approval Speed | 29% Faster |
| Distributor Performance Improvement | 37% |
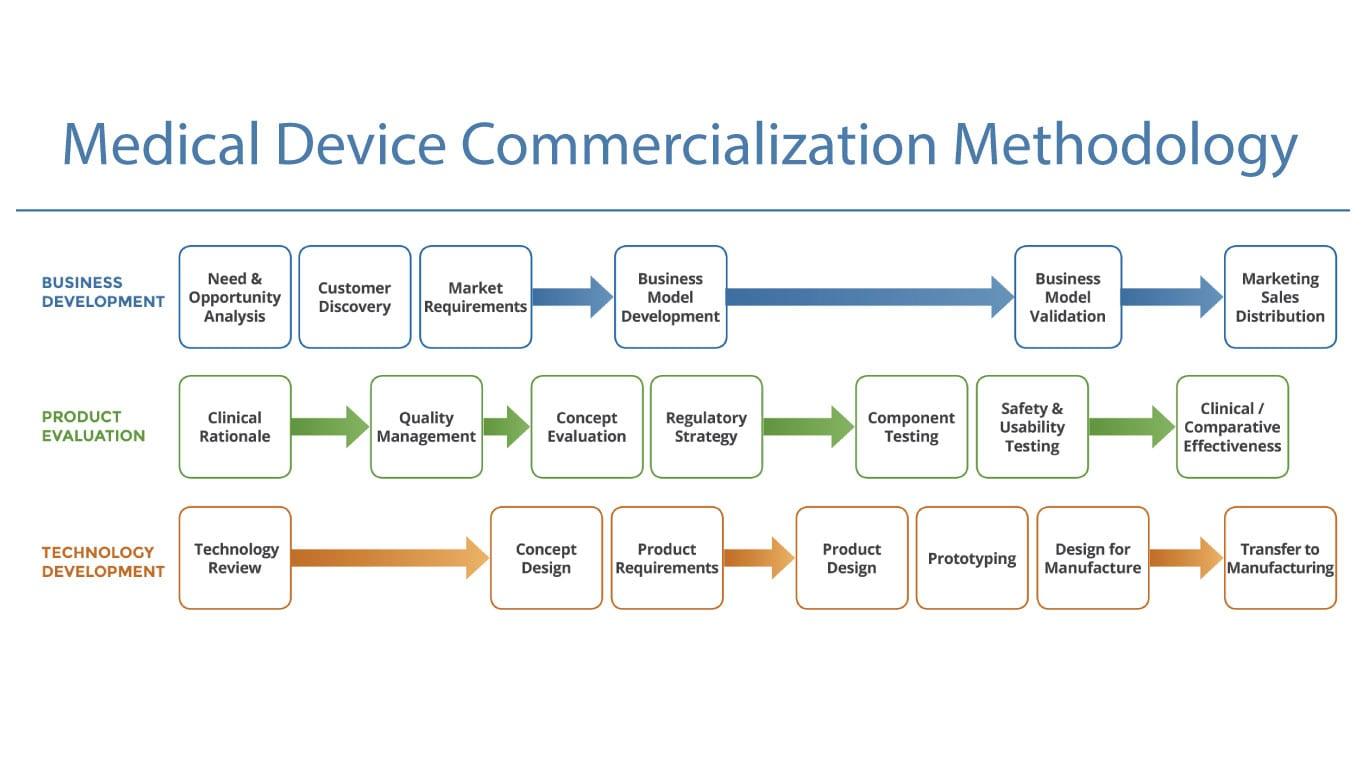
Strategic Recommendations
Short-Term Recommendations
- Prioritize high-reimbursement markets
- Build regulatory intelligence capabilities
- Establish regional commercialization hubs
Mid-Term Recommendations
- Develop integrated digital health ecosystems
- Strengthen hospital and payer collaborations
- Expand clinical evidence generation
Long-Term Recommendations
- Invest in AI-powered commercialization analytics
- Create strategic manufacturing partnerships
- Build scalable multi-market commercialization infrastructure
Conclusion
International MedTech commercialization requires more than product innovation. Companies must combine market intelligence, regulatory readiness, reimbursement planning, and strategic partnerships to succeed globally.
A structured market prioritization and partner strategy framework enables MedTech organizations to:
- Accelerate international expansion
- Improve commercialization ROI
- Reduce market-entry risk
- Enhance regulatory execution
- Build sustainable global growth
Organizations that align commercialization strategy with regional healthcare dynamics and strong partnership ecosystems will be best positioned to lead the next phase of global MedTech innovation.
Get Full Case Study
Enter deatils to get full case study.


